Simulations and experiments show that hydrogen bonds play an important role for the stability of peptide bonds against UV radiation.
In collaboration with the groups of Elsa Shanchez-Garcia (Mülheim) and Wolfram Sander (Bochum), Rachel Crespo-Otero and I have investigated the formation of weakly-bound dimers of N-methylformamide (NMF) and their photochemistry after UV irradiation.
The aim was to understand the effect of UV radiation on peptide bonds.
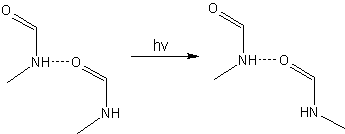
Starting from a trans(donor)-trans(acceptor) dimers, the experiments carried out in Sander’s group showed that the main products formed after irradiation are the trans(donor)-cis(acceptor) dimers.
Moreover, in contrast to the photochemistry of the NMF monomers [1], no dissociative products were observed after 248 nm irradiation of the NMF dimers.
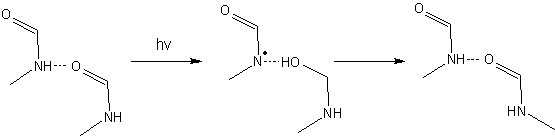
We have explained the absence of dissociative products using nonadiabatic dynamics simulations. These simulations showed that NMF dimers are protected by a proton-transfer mechanism in the excited state that is faster than the photo-dissociation.
The excited-state proton transfer occurs according the Sobolewski-Domcke mechanism [2], where an electron is transfered before and then followed by the proton. This process can be better seen in the movie below.
This movie shows the evolution of the electronic density difference between the excited and the ground states (dimer at left). Orange indicates electron-deficient regions, while green indicates electron-rich regions.
Although we have successfully explained the photostability issue, the theoretical results were not enough to explain why only the acceptor NMF monomer isomerized from trans to cis. This is a piece of mystery for further investigations.
But the discovery that the hydrogen bonding has such significant effect on the photochemical stability of NMF may have important implications to understand the stability of peptide-bonded systems to UV irradiation.
Reference
R. Crespo-Otero, A. Mardykov, E. Sanchez-Garcia, W. Sander, M. Barbatti, Photo-stability of peptide-bond aggregates: N-methylformamide dimers; Phys. Chem. Chem. Phys., doi:10.1039/C4CP02518K (2014).