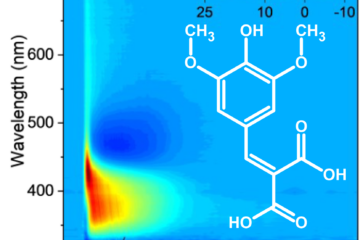
Methyl ferulate: protection efficiency with minimal photochemical degradation.
In brief:
- Conformational Dependency: This work demonstrated how molecular conformations influence Franck-Condon activity and UV absorption properties of methyl ferulate.
- Solvation Effects: It revealed that microsolvation induces redshifts and alters hydrogen-bonding interactions, stabilizing specific conformers.
- Excited-State Dynamics: We identified a long-lived excited state (~3 ns) and rapid triplet decay (~30 ns), optimizing UV protection with minimal photochemical degradation.
Our new paper, now available in the Journal of Physical Chemistry A, explores the spectroscopic and excited-state dynamics of methyl ferulate (MF), a naturally occurring UV-protective molecule. This study, an experimental-theoretical collaboration coordinated by Wybren Jan Buma at the University of Amsterdam, offers insights into how conformational flexibility and solvation influence its photophysical behavior, paving the way for designing next-generation UV filters and photothermal materials.
Methyl ferulate is part of the cinnamate family and is known for its strong UV absorption and applications in sunscreens. Using resonance-enhanced multiphoton ionization spectroscopy combined with advanced quantum chemical calculations, we mapped out MF’s absorption characteristics and decay pathways. The results revealed the dominant role of the bright ππ* state, which initiates the molecule’s photophysics.
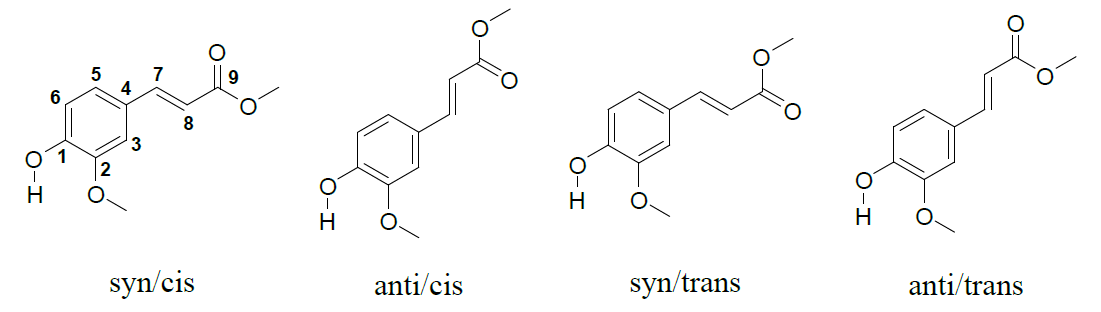
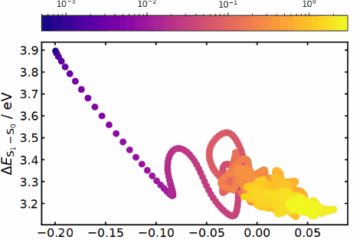
A key focus was understanding MF’s conformational dependency. Through experimental and theoretical Franck-Condon analysis, we identified that the molecular conformations significantly affect the observed vibrational activity. Solvation studies further highlighted the role of water in stabilizing specific conformations, with microsolvation leading to redshifts in UV absorption and new hydrogen-bonding interactions. These findings underscore the importance of the molecule’s microenvironment in its behavior.
MF’s excited-state dynamics also provided surprising insights. Unlike some cinnamate derivatives, MF shows a longer-lived excited state (~3 ns) before transitioning to a triplet state via intersystem crossing (ISC). From there, it undergoes a relatively fast (~30 ns) spin-forbidden decay to the ground state. This balance of lifetimes positions MF as a promising candidate for UV applications, combining protection efficiency with minimal photochemical degradation.
By dissecting these pathways with TDDFT and DFT/MRCI, we gained a quantitative understanding of the excited-state mechanisms in methyl ferulate. The insights presented in this study are vital for designing improved sunscreen molecules and expanding the use of cinnamate derivatives in photothermal materials.

For a deeper dive into the methods and implications of our work, check out the full paper in the Journal of Physical Chemistry A. This research marks another step toward bridging fundamental photochemistry with real-world applications in UV protection and sustainable materials.
MB
Reference
- I. Romanov, Y. Boeije, J. M. Toldo, M. T. Do Casal, M. Barbatti, W. J. Buma, Spectroscopy and Excited-State Dynamics of Methyl Ferulate in Molecular Beams, J. Phys. Chem. A (2024). 10.1021/acs.jpca.4c05792